Delivering Pharmaceutical Innovations Domestically and Globally
Toward Realizing the JPMA's Vision for 2035Our activities are centered on the three pillars of Innovation, Access, and Trust.
Consistently produce innovations to benefit health spans and the economy
Bring innovative new drugs to the public more quickly to enhance health security
Commit to ethical conduct and transparency to foster trust in the pharma industry
-
Latest JPMA Newsletter JPMA holds its first Town Hall Meeting! Creating the Future Together with the Power of Change and Challenge
-
Event Report: Updated April 23 "2025 Meeting of Compliance Managers and Practitioners in Charge of Compliance Management" was held.
-
DATA BOOK 2026" is now available.
-
Scientific and Practical Requirements for Drug Candidate Substances for Companies ~Statement of TPP
-
JPMA's Vision for 2035" and "JPMA's Policy Proposals for 2025" released on Feb. 26, 2025
Updates
-
Drug Evaluation Committee
Positioning Map Analysis of Patients' Web-based Drug Information Search" is now available.
-
Code Compliance Committee Event Report
The "FY2025 Meeting of Compliance Management Managers and Practitioners" was held.
-
Public Affairs Committee Event Report
Girls Meet STEM to Experience "The Moment Drugs are Created"-Girls in Junior High and High School Take on the World of Drug Discovery-.
-
News Release APAC Project
On the Occasion of the 15th APAC (Asia-Pacific Association of Pharmaceutical Manufacturers) Conference
-
News Release APAC Project
The 15th APAC Press Conference
-
Drug Evaluation Committee
A Beginner's Guide to Using NDB in the Pharmaceutical Industry" is now available.
-
News Release APAC Project
On the Occasion of the 15th APAC (Asia-Pacific Association of Pharmaceutical Manufacturers) Conference
-
News Release APAC Project
The 15th APAC Press Conference
-
News Release Drug Evaluation Committee
3 organizations have produced and released educational materials for the general public on "Don't believe everything you read" in the medical and health fields.
-
Announcement Public Affairs Committee
The JPMA website (top page) has been renewed.
-
Announcement Code Compliance Committee
Revision of Transparency Guideline for the Relation between Corporate Activities and Medical Institutions
-
Announcement
Business Policy, Business Plan, and Implementation Plan" for FY2026 is now available.
-
Drug Evaluation Committee
Positioning Map Analysis of Patients' Web-based Drug Information Search" is now available.
-
Code Compliance Committee Event Report
The "FY2025 Meeting of Compliance Management Managers and Practitioners" was held.
-
Public Affairs Committee Event Report
Girls Meet STEM to Experience "The Moment Drugs are Created"-Girls in Junior High and High School Take on the World of Drug Discovery-.
-
News Release APAC Project
On the Occasion of the 15th APAC (Asia-Pacific Association of Pharmaceutical Manufacturers) Conference
-
News Release APAC Project
The 15th APAC Press Conference
-
Drug Evaluation Committee
A Beginner's Guide to Using NDB in the Pharmaceutical Industry" is now available.
-
Code Compliance Committee Event Report
The "FY2025 Meeting of Compliance Management Managers and Practitioners" was held.
-
Public Affairs Committee Event Report
Girls Meet STEM to Experience "The Moment Drugs are Created"-Girls in Junior High and High School Take on the World of Drug Discovery-.
-
Intellectual Property Committee Event Report
Held "2026 Life Science IP Forum
-
Review Board of Ethical Drug Product Information Brochure Event Report
Workshop for Persons Responsible for Product Information Overview Management and Practical Responsibility for Product Information Overview Management in 2025" was held.
-
Public Affairs Committee Event Report
Report on the JPMA Media Forum
-
Pharmaceutical Industrial Policy Committee Event Report
JPMA Rare Disease Day 2026 Symposium" Report Video and Report are now available.
About JPMA
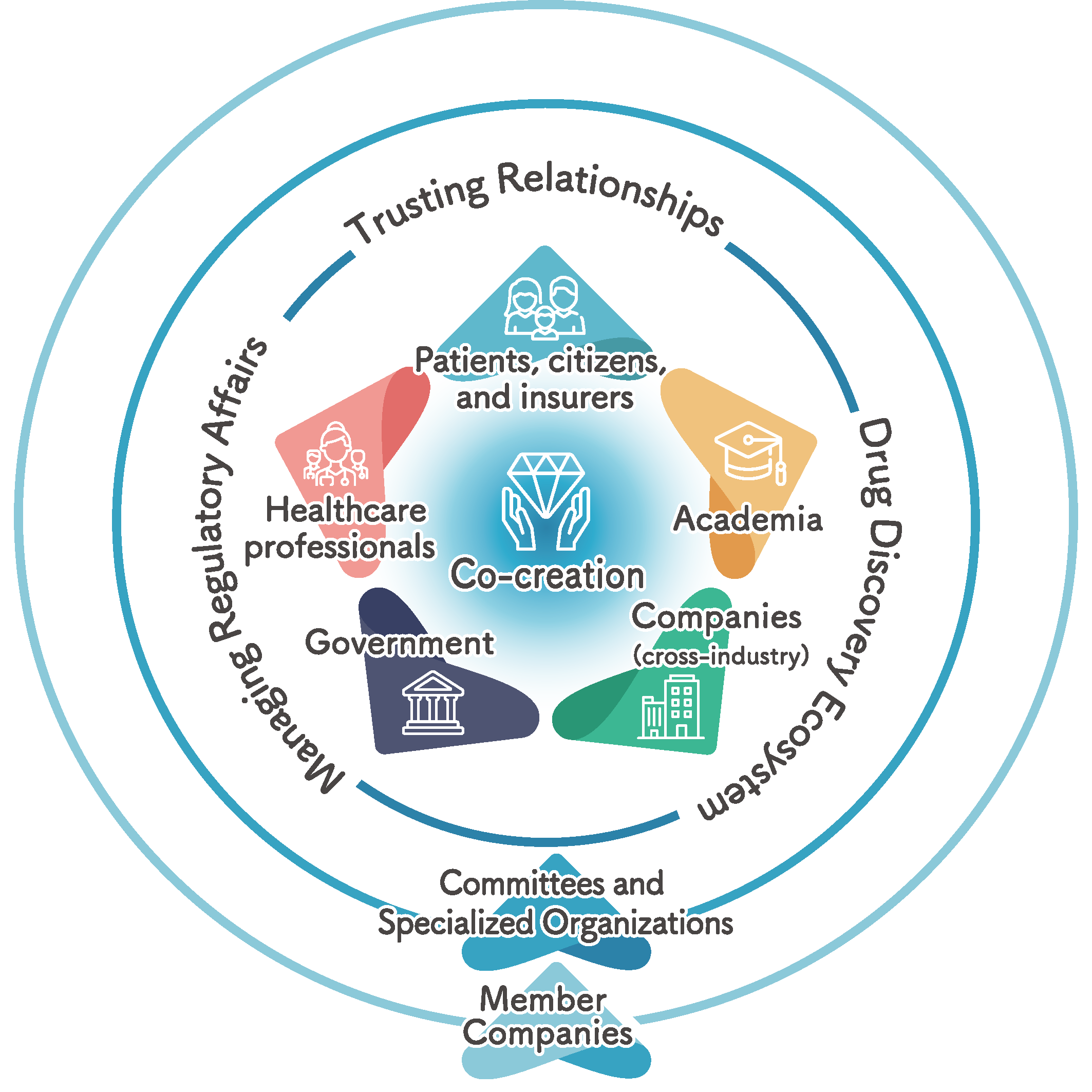
Under the JPMA Board of Directors, 12 committees composed of member companies and 6 specialized organizations, including the Office of Pharmaceutical Industry Research (OPIR) which functions as a think tank, carry out a wide range of initiatives. These activities aim to establish a robust drug discovery ecosystem for the development of innovative pharmaceuticals, ensure compliance with regulatory frameworks such as the Pharmaceuticals and Medical Devices Act, and build trust with key stakeholders.
Each committee and specialized body bring together a diverse pool of talent - from young professionals to experienced experts - working across organizational boundaries. Through mutual collaboration and constructive exchange, they advance pharmaceutical innovation, promote the appropriate use of medicines, enhance transparency in distribution, foster international cooperation, and formulate policy proposals. In addition, JPMA conducts various surveys and research projects to address industry-wide challenges. By working closely with government authorities, patient advocacy groups, and other relevant organizations, JPMA supports the sound and sustainable development of the pharmaceutical industry.
JPMA's Vision
Consistently produce innovations to benefit health spans and the economy
- Pharmaceutical innovations
- Leading-edge scientific technology
- Pharmaceutical manufacturing technology
- Domestic manufacturing infrastructure
- Making Japan a science and technology powerhouse
- Fostering highly skilled talent
- PPI/E
- Drug discovery that involves patients and the public
- Co-creation
- Drug discovery ecosystem
- Job creation
- Extending health spans
- Well being
- Pharma as a core industry
- Economic growth
Bring innovative new drugs to the public more quickly to enhance health security
- A social security system that gives patients swift access to “the latest treatments” and stable access to “global-standard treatments”
- Positive investment cycle
- Building an attractive domestic market
- Drug pricing that properly factors in the worth of innovation
- Health security for Japan
Commit to ethical conduct and transparency to foster trust in the pharma industry
- Sustainable Development Goal 3 (Ensure healthy lives and promote well-being for all at all ages)
- Overcome the social challenges raised by Japan’s aging population
- Ethical soundness, code of behavior, and transparency
- Boost public literacy in health and medicine
- Social significance of pharmaceuticals and the pharma industry
- Encourage the public and younger people to enter the pharma industry
- Climate change
- Environmental issues
Member Companies
The JPMA has 68 R&D-oriented JPMA member companies (as of April 1, 2026).













































