It is said that it takes an average of 9 to 17 years for a new drug to reach patients through numerous stages, including the discovery of a new substance that will serve as the basis for the drug, basic research, non-clinical testing, clinical testing, and review. And to support such long years of research and development, large amounts of R&D funds are required.
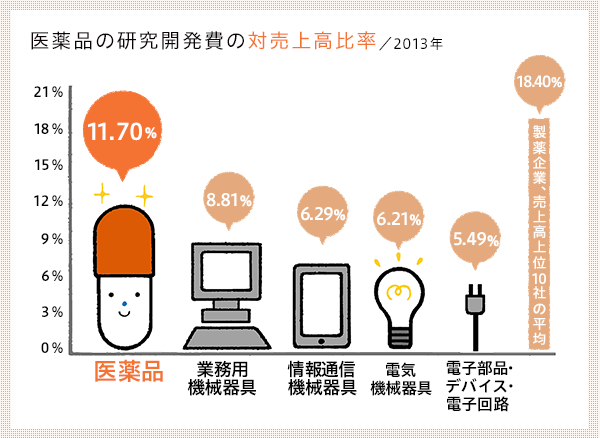
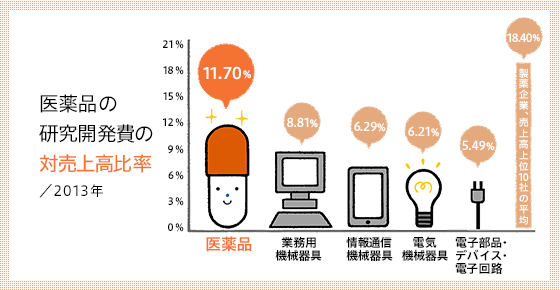
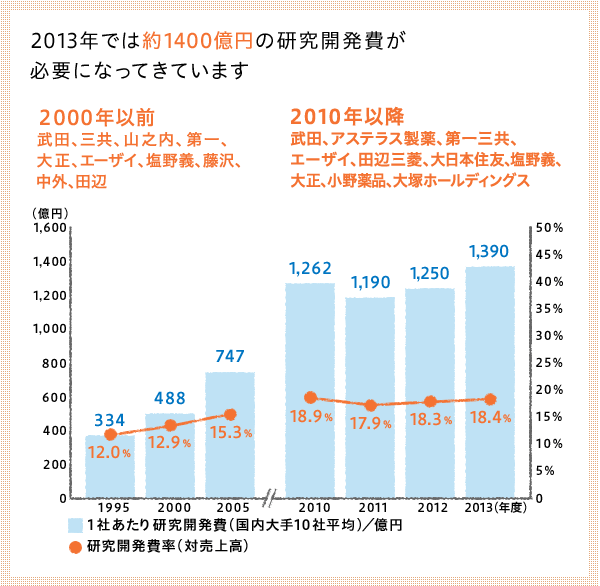
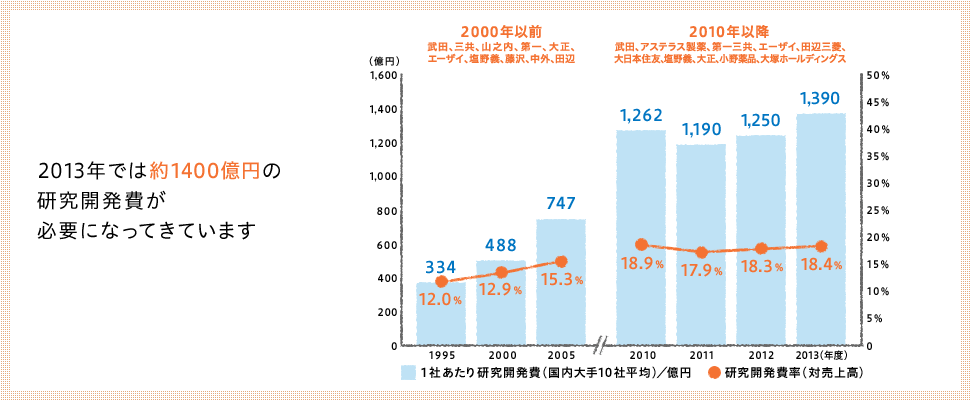
Japan's commitment to the creation of new drugs is also reflected in its R&D ratio. While the ratio of R&D to sales in the Japanese manufacturing industry is 4.15%, the ratio in the pharmaceutical manufacturing industry is 11.70%, and the average for the 10 largest pharmaceutical companies is 18.40%.
Why invest such large amounts of money in R&D? It is because they believe that a single new drug can bring smiles to the faces of countless patients.