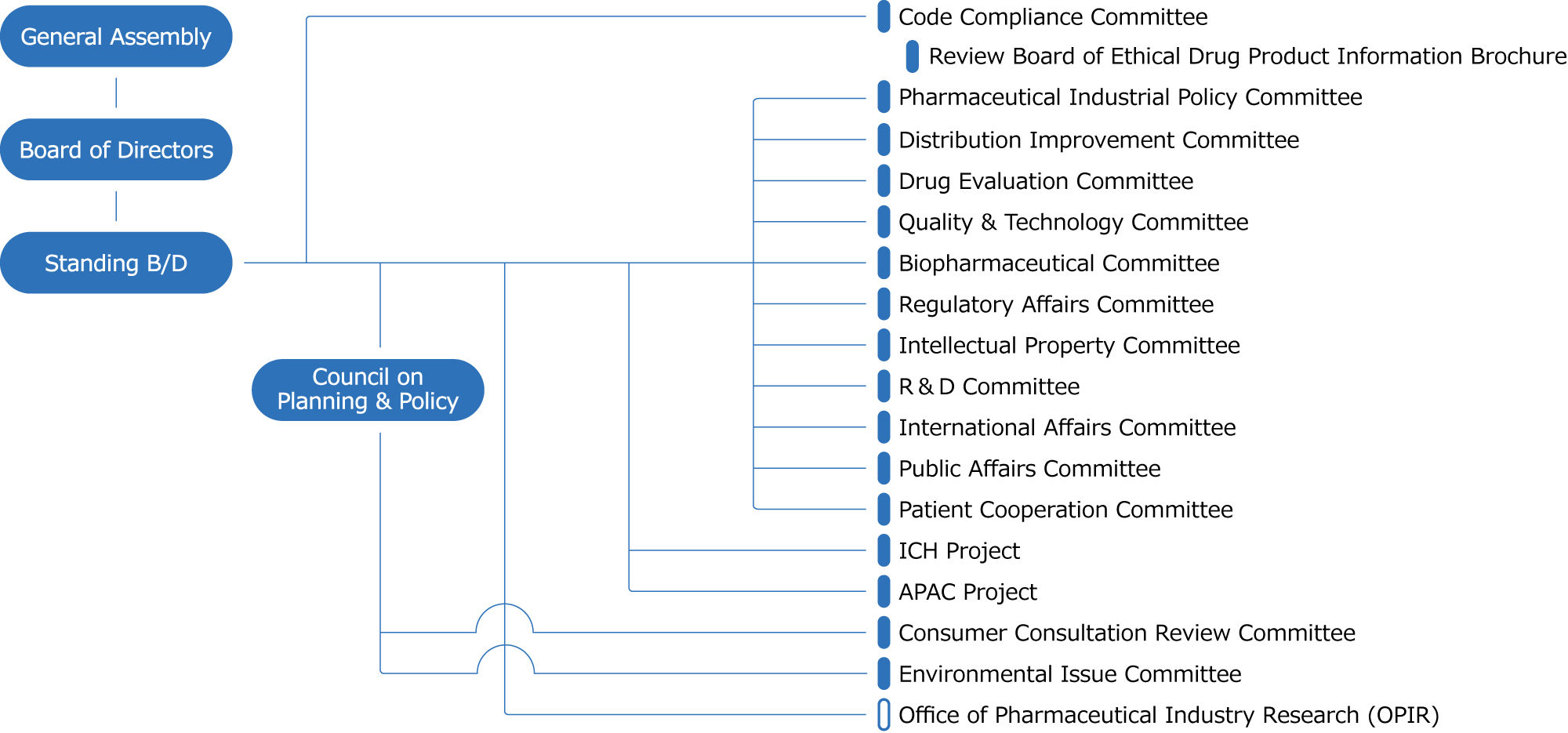
| Code Compliance Committee | Promotes compliance among JPMA member companies, administers the JPMA Code of Practice, and supports member companies' code compliance promotion activities. |
| Review Board of Ethical Drug Product Information Brochure | The committee independently reviews product information summaries of ethical drugs and promotional materials such as advertisements in specialized magazines prepared by member companies to ensure the proper use of ethical drugs. |
| Pharmaceutical Industrial Policy Committee | The Pharmaceutical Industrial Policy Committee examines issues and makes policy proposals concerning industrial promotion, innovation promotion, taxation, elimination of tariffs on pharmaceuticals*, and other issues that cannot be addressed by the standing committees in order to respond promptly to changes in the environment surrounding the pharmaceutical industry.
|
| Distribution Improvement Committee | The JPMA works to resolve various issues and makes policy recommendations through consultations with related organizations and government agencies in order to realize more transparent and efficient distribution of pharmaceuticals. |
| Drug Evaluation Committee | In order to promote pharmaceutical research, development, post-marketing safety measures/appropriate use, and medical affairs activities, the committee examines technologies and regulations for each of these activities, makes policy recommendations, and conducts educational activities. |
| Quality & Technology Committee | The Quality Committee investigates and researches pharmaceutical GMP, manufacturing and analytical technologies, and related issues from an international perspective, and establishes and actively promotes measures to improve the reliability of pharmaceutical quality, thereby contributing to the improvement of medical care and the sound development of the industry. |
| Biopharmaceutical Committee | The Biopharmaceuticals Committee makes policy recommendations regarding the development of infrastructure for the promotion of biopharmaceutical research and development. It also conducts surveys and research on various technical issues related to biopharmaceuticals, from research and development to production and post-marketing activities, and makes recommendations for improvement to the government and other relevant agencies. |
| Regulatory Affairs Committee | The committee studies and examines various issues related to the Pharmaceuticals and Medical Devices Act, and pharmaceutical regulations concerning approval screening and safety measures, etc., and makes policy recommendations on various pharmaceutical administration matters, such as improving the efficiency of corporate activities and expediting approval screening, from the standpoint of R&D-oriented pharmaceutical companies. |
| Intellectual Property Committee | In order for Japan's pharmaceutical industry to maintain its competitiveness in the international economic community and to continue to develop, the committee works with relevant ministries, agencies, and other organizations to actively make policy recommendations regarding intellectual property systems not only in Japan but also around the world. The committee also disseminates information to promote understanding of intellectual property issues in the life science field. |
| R & D Committee | The committee aims to realize effective drug discovery R&D for member companies by collecting and sharing information on priority issues related to non-clinical and clinical trials from the research stage, making policy recommendations to the Japan Agency for Medical Research and Development (AMED) and ministries, and promoting open innovation. |
| International Affairs Committee | The International Committee deepens cooperation with regulatory authorities, pharmaceutical organizations, and the International Federation of Pharmaceutical Manufacturers and Associations (IFPMA) in various countries from the three perspectives of international expansion, international harmonization, and contribution to global health, and works to resolve international issues faced by the industry and member companies. |
| Public Affairs Committee | The committee develops activities to raise awareness and promote correct understanding of pharmaceuticals and the pharmaceutical industry. It also promotes proactive public relations activities to realize the policy proposals advocated by JPMA. |
| Patient Cooperation Committee | The committee actively exchanges opinions with patient groups to promote mutual understanding, discusses initiatives from the patients' perspective, and promotes better collaboration with patient groups. In addition, the committee will make efforts to disseminate the "Transparency Guideline for the Relationship between Corporate Activities and Patient Organizations". |
| ICH Project | JPMA participates in the International Council for Harmonisation of Technical Requirements for Pharmaceuticals for Human Use (ICH) and contributes to faster and more efficient drug development through the development of harmonized guidelines. We also organize ICH Public Meetings and Guideline Workshops to share information and raise awareness. |
| APAC Project | The Asian Pharmaceutical Association Council (APAC) is a platform of 14 research-based pharmaceutical organizations in Asia to identify issues and make recommendations toward the realization of the mission of "bringing innovative medicines to the people of Asia in a timely manner. The APAC aims to create results to achieve the mission through discussions with government and academia on the defined themes through studies and deliberations by each of the related working groups and task forces. |
| Consumer Consultation Review Committee | The committee will contribute to "patient-participatory medicine" by studying and making proposals on the provision of drug information to the public and the ideal corporate consultation service for pharmaceuticals, in order to improve the reliability of the information provided and the recognition of the service as a consultation service. |
| Environmental Issue Committee | The committee will study measures to promote measures to deal with various issues related to global environmental protection, with the aim of promoting environmental protection activities. |