Topics ICH Fukuoka Meeting Held in Fukuoka, Japan
The International Council for Harmonisation of Technical Requirements for Pharmaceuticals for Human Use (ICH) held its meeting in Fukuoka, Japan, from June 1 to 5, 2024, the first time in six years since the ICH Kobe meeting in June 2018.
 Overall photo of the Assembly
Overall photo of the Assembly
In addition to the Assembly, which was attended by all ICH members, the Management Committee and other meetings were held to prepare for the discussions to be held at the Assembly and to consider the management of the ICH. Thirteen expert working groups (working groups) also met in parallel during the same period to discuss ICH technical guidelines and to make progress on specific guideline documents. The ICH meetings are held twice a year, with on-site participation as a general rule.
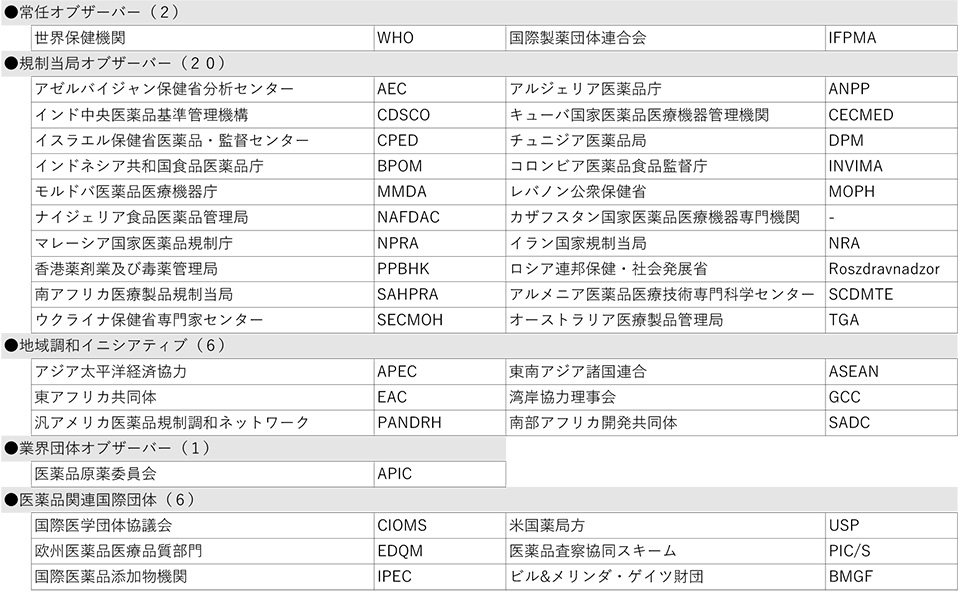
The breakdown of participating organizations at the Fukuoka meeting was as follows: 6 industry-government organizations*1 from Japan, the U.S., and the EU, which are founding members; 2 Standing Members (Health Canada and Swissmedic); 15 Members*2; 2 Standing Observers*3; and 17 Observers. More than 500 people from all over the world attended the Fukuoka meeting, and 35 JPMA members, including experts from the Working Groups, participated in the Fukuoka meeting.
The following is a list of special notes based on the ICH Management Committee and ICH Assembly at the Fukuoka meeting.
-
1U.S. Food and Drug Administration (FDA), European Commission/European Medicines Agency (EC/EMA), Ministry of Health, Labour and Welfare/Pharmaceuticals and Medical Devices Agency (MHLW/PMDA), Pharmaceutical Research and Manufacturers of America (PhRMA), European Federation of Pharmaceutical Manufacturers and Associations (EFPIA), Japan Pharmaceutical Manufacturers Association (JPMA)
-
2Brazilian Health Regulatory Agency (ANVISA), Argentina National Superintendence of Medicines, Food and Medical Technology(ANMAT), Mexican Federal Commission for Protection against Sanitary Risks (COFEPRIS), Egyptian Drug Agency (EDA), Health Sciences Authority of Singapore (HAS), Jordan Food and Drug Administration (JFDA), Korea Food and Drug Safety Administration (MFDS), UK Medical Products Regulatory Agency (MHRA), National Medical Products Administration (NMPA) of China, Saudi Food and Drug Administration (SFDA), Taiwan Food and Drug Administration (TFDA), Turkish Medicines and Medical Devices Agency (TITCK), Biotechnology Innovation Organization (BIO), Global Self-Care Federation (GSCF), International Generic and Biosimilar Medicines Association (IGBA)
-
3International Federation of Pharmaceutical Manufacturers and Associations (IFPMA), World Health Organization (WHO)
1. new members and observers
The Argentine authorities (Argentine National Administration of Drugs, Food and Medical Devices, ANMAT) and the Jordanian authorities (Jordan Food and Drug Administration, JFDA) have applied to become new ICH members. There were no new applications for ICH observer membership.
As a result, ICH now has 23 member organizations and 35 observer organizations, bringing the total number of ICH members to 58 (see the reference material at the end of this document).
2. trends in ICH technical topics
Working Groups with Meetings
A total of 13 working groups on 13 topics met in Fukuoka. Each working group discussed intensively for four or five days to make progress in the development of the guidelines.
| • | Q1/Q5C EWG: Revision of Stability Testing Guidelines for Drugs |
| • | Q2 (R2)/Q14 EWG: Revision of "Validation of Analytical Procedure" and "Analytical Procedure Development" |
| • | Q3E EWG: Evaluation and Control of Extractables and Leachables of Pharmaceutical and Biological Products |
| • | Q5A (R2) IWG: Revision of "Quality of Biopharmaceuticals: Viral Validation |
| • | Q6(R1) Informal WG: Revision of the Specifications Guidelines |
| • | Q9(R1) IWG: Revision of "Guidelines for Quality Risk Management" Q9(R1) IWG: Revision of "Quality Risk Management |
| • | E6 (R3) EWG: Revision of "Good Clinical Practice" |
| • | E20 EWG: Adaptive Clinical Trials |
| • | E21 informal WG: Inclusion of pregnant and breastfeeding individuals in clinical trials |
| • | M4Q(R2)EWG: Revision of the Common Technical Document for the Registration of Pharmaceuticals for Human Use: Quality |
| • | M11 EWG: Clinical electronic Structured Harmonised Protocol (CeSHarP) |
| • | M13 EWG: Bioequivalence for Immediate-Release Solid Oral Dosage Forms |
| • | M15 EWG: General Principles for Model-Informed Drug Development |
Topics that reached important milestones
No topics reached a step transition during the Fukuoka meeting, but the following topics reached important milestones in ICH in the six months since the November 2023 ICH Prague meeting.
| • | M12: Drug Interaction Study (reached Step 4 in May 2024) |
| • | M14:General Principles on Planning, Designing, Analysing, and Reporting of Non-interventional Studies That Utilise Real-World Data for Safety Assessment of Medicines (Reached Step 2 in May 2024) |
| • | E2D(R1): Post-Approval Safety Data: Definitions and Standards for Management and Reporting of Individual Case Safety Reports(Reached Step 2 in February 2024) |
Reaching Step 2 means that the draft guideline based on the technical document has been approved by the Assembly's regulatory representatives and will now move to Step 3, where regional and national regulatory authorities will conduct public comments. Reaching Step 4 means that the draft guidelines have been finally agreed upon and adopted by the Assembly's regulatory representatives, and the English version of the guidelines has been finalized. The implementation process by regional and national regulatory authorities will now begin.
3. Election of ICH Management Committee Members
The ICH Management Committee consists of Founding Members (3 from regulators and 3 from industry), Standing Members (2 from regulators), Standing Observers (WHO and IFPMA), and Elected Representatives (4 from regulators and 2 from industry). Elected Representatives are elected every three years, and the election was held this time. As a result of the election, the following organizations were selected as Elected Representatives to the Management Committee
| Regulatory Authority: | Brazilian Health Regulatory Agency (ANVISA), Korea Food and Drug Safety Administration (MFDS), National Medical Products Administration (NMPA), Saudi Food and Drug Administration (SFDA) |
| Industry: Biotechnology Innovation Organization (BIO), International Generic and Biosimilar Medicines Association (IGBA) | |
4. Adoption of New Topics
The following topics were adopted as new topics for future guideline development. The specific timing of the start of activities will be discussed in the future.
| • | ICH M7 Guideline Addendum "Risk Assessment and Control of Nitrosamine Impurities" |
5. Adoption of ICH Reflection Paper
At the Fukuoka meeting, a revised version of the reflection paper on "International harmonization of terminology for Real World Evidence (RWE) focusing on the effectiveness of medicinal products and integration of general principles for the design and reporting of studies using RWD" was reviewed and adopted by ICH Assembly. 6.
6. next ICH meeting
The next meeting of the ICH will be held in Montreal, Canada, November 2-6, 2024.
ICH actively discloses information on ICH activities, including the outcomes of ICH meetings, in order to deepen understanding of ICH activities among the general public as well as related parties. The results of the ICH Fukuoka meeting, concept papers on each topic, work plans, etc. are available on the ICH website ( https://www.ich.org/ ).
[Reference: List of ICH Members and Observers (as of June 2024)
Table 1 Members (23 organizations)
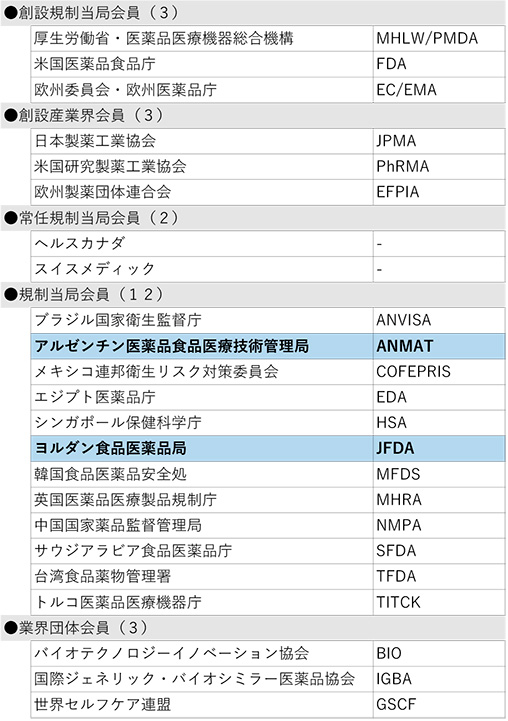
(Shaded areas in light blue were added at the ICH Fukuoka meeting.
Table 2 Observers (35 organizations)
( Mariko Kato, Director, International Regulatory Coordination Department)

