Topics ICH Incheon Meeting" Held
The International Council for Harmonisation of Technical Requirements for Pharmaceuticals for Human Use (ICH) held its second half of 2022 in Incheon, Korea, from November 12 to 16. Although the meeting was held in principle as a face-to-face meeting, online participation was available for participants who had difficulty in traveling due to the new coronavirus infection (COVID-19).
 Scene of the Meeting
Scene of the Meeting
The meeting included the preparation of proposals to be made and discussed at the Assembly, the Management Committee, which is responsible for the management of the ICH, and the Assembly, which is attended by all members. Discussions of the 10 expert working groups (working groups) that examine technical guidelines were also held in parallel at the same time to make progress in the preparation of ICH guidelines. At this Assembly, the Q13 Continuous Manufacturing of Drug Substances and Drug Products reached Step 4. Although the size of the meeting is smaller than in the past, we are gradually returning to the operation of ICH meetings prior to the COVID-19 epidemic.
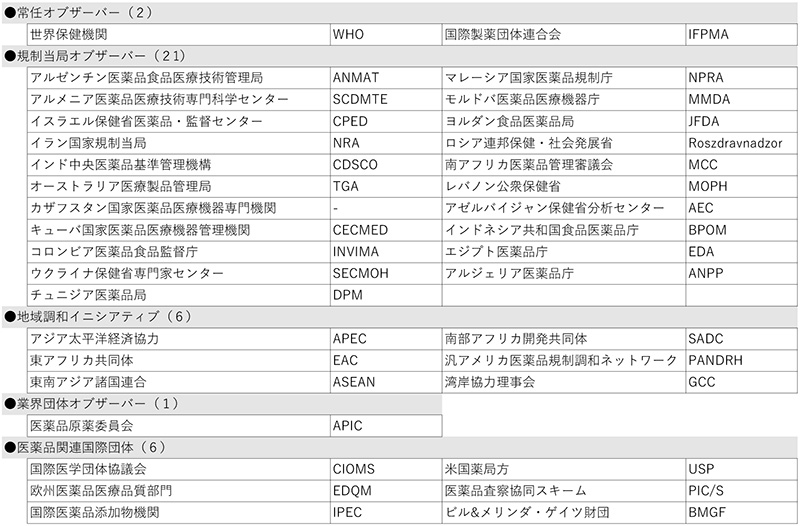
The Incheon meeting was attended by 24 organizations, including 6 industry-government organizations, 2 Standing Members, 12 Members, 2 Standing Observers, and other observers from Japan, the U.S., and the EU, all of which are founding members. More than 280 participants from around the world attended the local Incheon meeting, including 24 from JPMA.
The following is a list of special notes from the Incheon meeting.
1. New observers
The Directorate of Pharmacy and Medicine (DPM) Tunisia was approved as a new observer to ICH. As a result, the number of ICH members and observers increased by 20 and 1, respectively, to 36 organizations, bringing the total number of ICH organizations to 56 (see the reference material at the end of this document).
2. Trends in ICH Technical Topics
At the Incheon meeting, working groups on a total of 10 topics held active discussions and made progress.
Expert working groups with meetings
•Q1/Q5C informal WG:Targeted Revisions of the ICH Stability Guideline Series
•Q3E EWG: Guideline for Extractables and Leachables
•Q9(R1) EWG: Quality Risk Management
•Q13 EWG:Continuous Manufacturing of Drug Substances and Drug Products
•E2D(R1) EWG: Post-Approval Safety Data: Definitions and Standards for Management and Reporting of Individual Case Safety Reports
•E6(R3) EWG: Revision of "Guideline for Good Clinical Practice"
•E11A EWG: Pediatric Extrapolation
•M4Q(R2): Revision of "the Common Technical Document for the Registration of Pharmaceuticals for Human Use: Quality"
•M13A EWG: Bioequivalence for Immediate-Release Solid Oral Dosage Forms
•M15 informal WG: General Principles for Model-Informed Drug Development
Q1/Q5C and M15 will be new working groups established in 2022.
Step 4 reached
The Assembly reported the achievement of Step 4 for Q13 Continuous Manufacturing of Drug Substances and Drug Products. It will now enter the process of implementation at the respective regulatory authorities.
In addition, the Management Committee and Assembly also discussed progress management of all existing topics. The following topics were reported to have reached important milestones since the last ICH Athens meeting
| • | E19: A Selective Approach to Safety Data Collection in Specific Late-Stage Pre-Approval or Post-Approval Clinical Trials (Step 4 reached in September 2022) (Step 4 reached) |
| • | S1B (R1): Addendum to the Guidelines on Testing for Carcinogenicity Studies of Pharmaceuticals" (Reached Step 2 in September 2022) |
Step 2 reached
The following topics will be subject to public comment in each country/region
| • | M11: Clinical electronic Structured Harmonized Protocol (CeSHarP) (Reached Step 2 in September 2022) |
| • | Q5A(R2): Viral Safety Evaluation of Biotechnology Products Derived from Cell Lines of Human or Animal Origin(Step 2 will be reached in September 2022) |
3. ICH Awards
The ICH Award, which recognizes experts who have contributed to the study of the ICH Guidelines, was launched in 2022, and 12 people received the award in 2022. From Japan, three winners from the regulatory side and two winners from the industry side were selected for their important leadership and sustained contributions to the ICH working groups. The first two recipients from the JPMA were Yasushi Komiyama, Vice Chairperson of the Drug Evaluation Committee's Data Science Subcommittee, and Tomonori Nakagawa, Chairperson of the Quality & Technology Committee's ICH Quality Group.
4. next ICH meeting
The next ICH meeting will be held in Vancouver, Canada from June 9 to 13, 2023. The next meeting is also scheduled to be face-to-face.
ICH actively discloses information on ICH activities, including the outcomes of ICH meetings, to deepen understanding of ICH activities among the public as well as related parties. The results of the ICH Incheon Meeting, concept papers on each topic, and the work plan are available on the ICH website ( https://www.ich.org/ ).
[Reference: List of ICH Members and Observers (as of November 2022)
Table 1 Members (20 organizations)
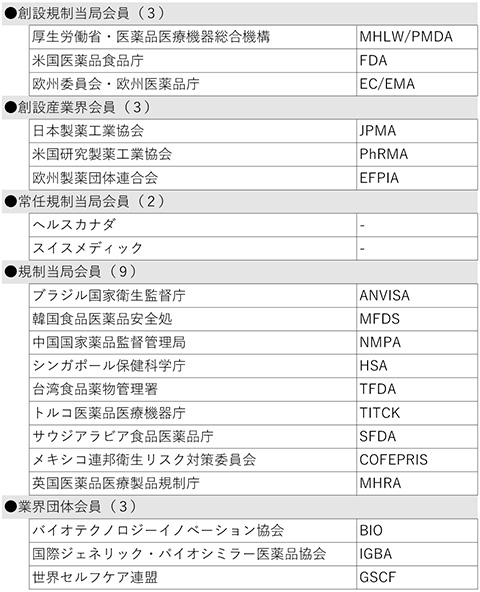
Table 2 Observers (36 organizations)
( Mariko Kato, Director, International Regulatory Coordination Department)

